
FDA Observations in Dietary Supplements: Trends and Part 111 Compliance Risks
FDA observations in dietary supplements continue to highlight recurring compliance gaps under 21 CFR Part 111. Recent Form 483 data shows that specification-related deficiencies remain the most common findings during inspections of supplement manufacturers.
At cGMP Consulting, we work directly with dietary supplement manufacturers responding to FDA inspections, addressing Form 483 findings, and strengthening compliance systems under 21 CFR Part 111. The latest FDA data reinforces a clear pattern that manufacturers cannot afford to ignore.
Last year we shared Top FDA Observations in Dietary Supplement Manufacturing; this article aims to educate you on these trends and offers practical tips for avoiding these findings in your operations and help you understand the dietary supplement regulations.
FDA Trends
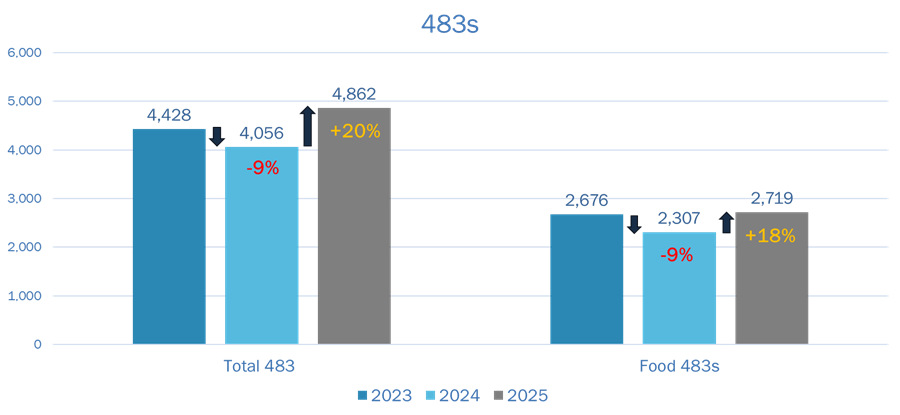
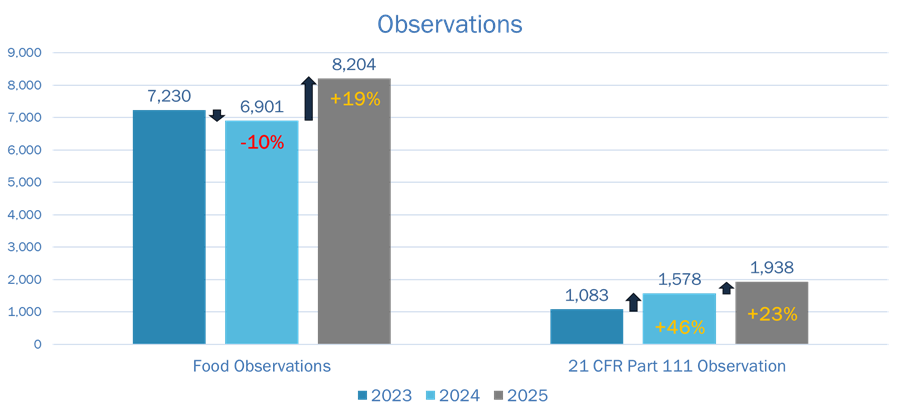
FDA inspection activity trends upward, particularly when it comes to Part 111.
- Total FDA Form 483s issued in 2025: 4,862
- Food-related 483s in 2025: 2,719 (56% of total)
- Total observations from Food 483s: 8,204
- Part 111-specific observations: 1,938
Dietary supplements fall under the Food category, but Part 111 observations are increasing at a faster rate than the overall Food category. This signals increased scrutiny of dietary supplement cGMP and dietary supplement manufacturing compliance systems.
Top Two Part 111 Observations
1. Failure to Establish Finished Product Specifications
“You did not establish product specifications for identity, purity, strength, and composition of a finished dietary supplement.”
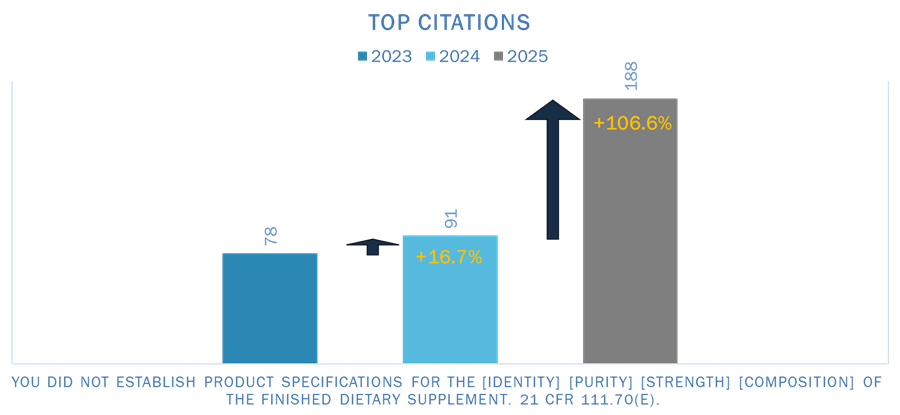
FDA continues to emphasize that specification development is a foundational requirement under dietary supplement GMP requirements.
Under 21 CFR Part 111 compliance, manufacturers must establish written specifications to ensure finished products:
- Contain the ingredients declared on the label
- Deliver the labeled strength and composition
- Meet purity and quality standards
- Remain consistent across batches
Without approved specifications, batch release decisions lack objective criteria, creating significant GMP compliance dietary supplements risks.
2. Failure to Establish Identity Specifications for Each Component
“You did not establish an identity specification for each component.”
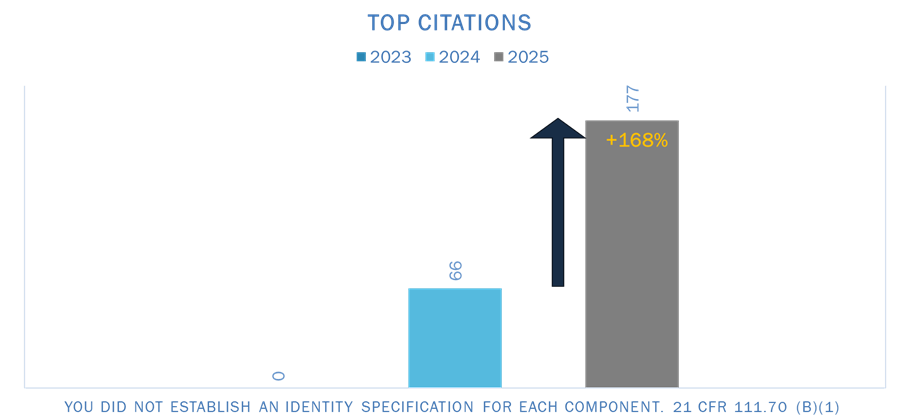
This observation appeared 66 times in 2024 after zero occurrences the year before, signaling a clear emphasis in inspection focus.
Under dietary supplement cGMP, manufacturers must:
- Establish written identity specifications for every component
- Perform at least one appropriate test or examination to verify identity
- Approve components before use and the suppliers who provide them
Overreliance on supplier Certificates of Analysis without proper qualification remains a common finding during dietary supplement FDA inspections.
Why Specifications Are a Core Regulatory Focus
Current dietary supplement manufacturing trends include:
- Increased use of botanical and novel ingredients
- Global sourcing of raw materials
- Complex, multi-ingredient formulations
- Accelerated product launch timelines
These trends increase variability and risk. Under 21 CFR Part 111, manufacturers must establish specifications for:
- Incoming components
- In-process materials
- Finished products
- Packaging and labeling
Common compliance gaps identified during FDA inspections include:
- Missing or incomplete written specifications
- Unvalidated or poorly controlled test methods
- Weak supplier qualification programs
- No defined linkage between specifications and batch release decisions
Strong supplement quality systems integrate specifications, testing, documentation, and product disposition into a single controlled process.
How to Strengthen Your Compliance Framework
To prevent recurring observations, manufacturers should:
- Develop complete written specifications for identity, purity, strength, composition, and contamination limits.
- Implement structured supplier qualification programs with defined CoA verification procedures.
- Use validated laboratory test methods.
- Ensure batch release decisions are systematically linked to approved specifications.
- Conduct routine internal audits to strengthen dietary supplement manufacturing compliance.
Compliance as a Competitive Advantage
Modern dietary supplement trends show that retailers and regulators expect transparency and robust quality oversight. Companies that invest in strong dietary supplement cGMP systems are better positioned to:
- Reduce enforcement risk
- Maintain retailer relationships
- Support market expansion
- Successfully navigate dietary supplement FDA inspections and customer audits
Proactive compliance protects your brand and supports sustainable growth by avoiding time consuming observations 483s.
How cGMP Consulting Can Help
cGMP Consulting partners with manufacturers to strengthen:
- Specification development
- Supplier qualification programs
- Risk-based testing strategies
- Full 21 CFR Part 111 compliance systems
- Audit and inspection readiness
We are ISO/IEC 17020 accredited to provide cGMP Certification and offer independent, expert evaluations of your supplement quality systems.
If you want to align your operations with current dietary supplement manufacturing trends while minimizing regulatory risk, contact our team today and schedule a call.
Source:
U.S. Food and Drug Administration (FDA). Inspection Observations Database.
https://www.fda.gov/inspections-compliance-enforcement-and-criminal-investigations/inspection-references/inspection-observations
Accessed February 20, 2026.





